

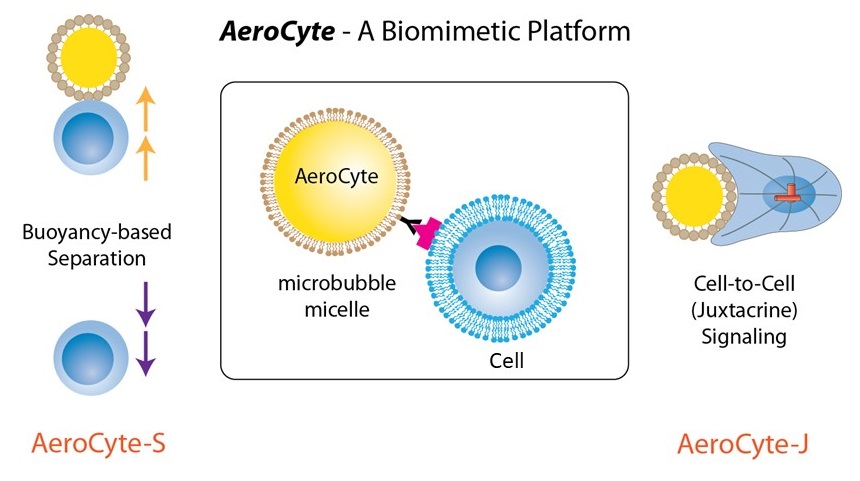
AeroCytek, Inc., the successor-in-interest to the R&D-focused Diagnologix, LLC in 2024, specializes in phospholipid microbubble-based cell processing to address manufacturing bottlenecks in clinical applications, offering enhanced capabilities and a broader market reach.
AeroCytek creatively applies FDA-approved phospholipid-shelled microbubbles for reversible buoyancy-based cell sorting and as tunable artificial cells. These microbubbles have been employed to select, activate, and transduce T cells for ex vivo CAR-T cell production in a preclinical animal model. They can dissolve in solution instantly by increasing ambient pressure or spontaneously over a longer period. Functionally, these cell-like microbubbles (designated AeroCyte) can be categorized by their activities with cells into three groups:
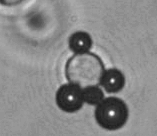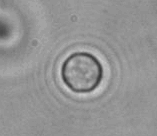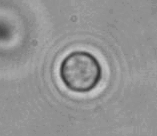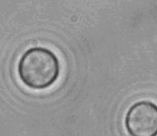
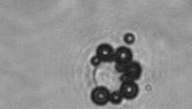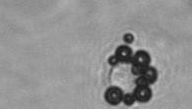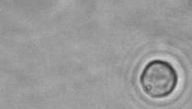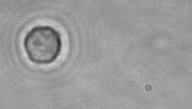
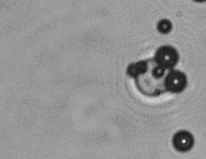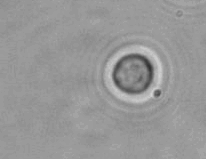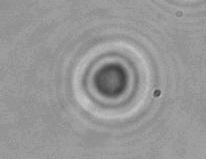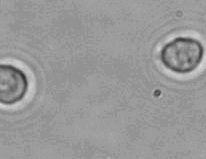
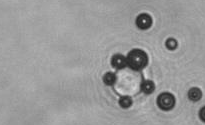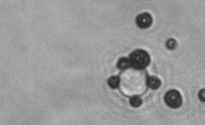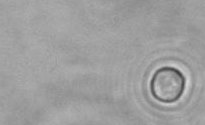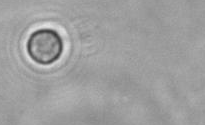
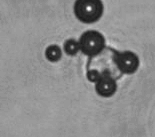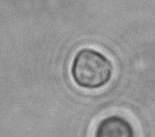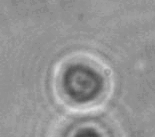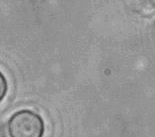
The dissolution of cell-bound lipid-shelled microbubbles occurs due to air compression at 2 atm, as depicted in a series of time-lapse images captured over 2 seconds.
In addition to clinical development, AeroCytek currently offers research-use-only (RUO) products under contract. GMP-grade products are under development. Please contact the AeroCytek team (contact@aerocytek.com) for sales, partnerships, and other information.
AeroCytek has been awarded PHS-funded research and has implemented a Financial Conflict of Interest (FCOI) Policy in accordance with the Federal Department of Health and Human Services regulations on Promoting Objectivity in Research.